Dr Clare Materia Medica
Introduction to the Dispensing of Dr Clare’s Blended Herbs
Special | A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | ALL
F |
|---|
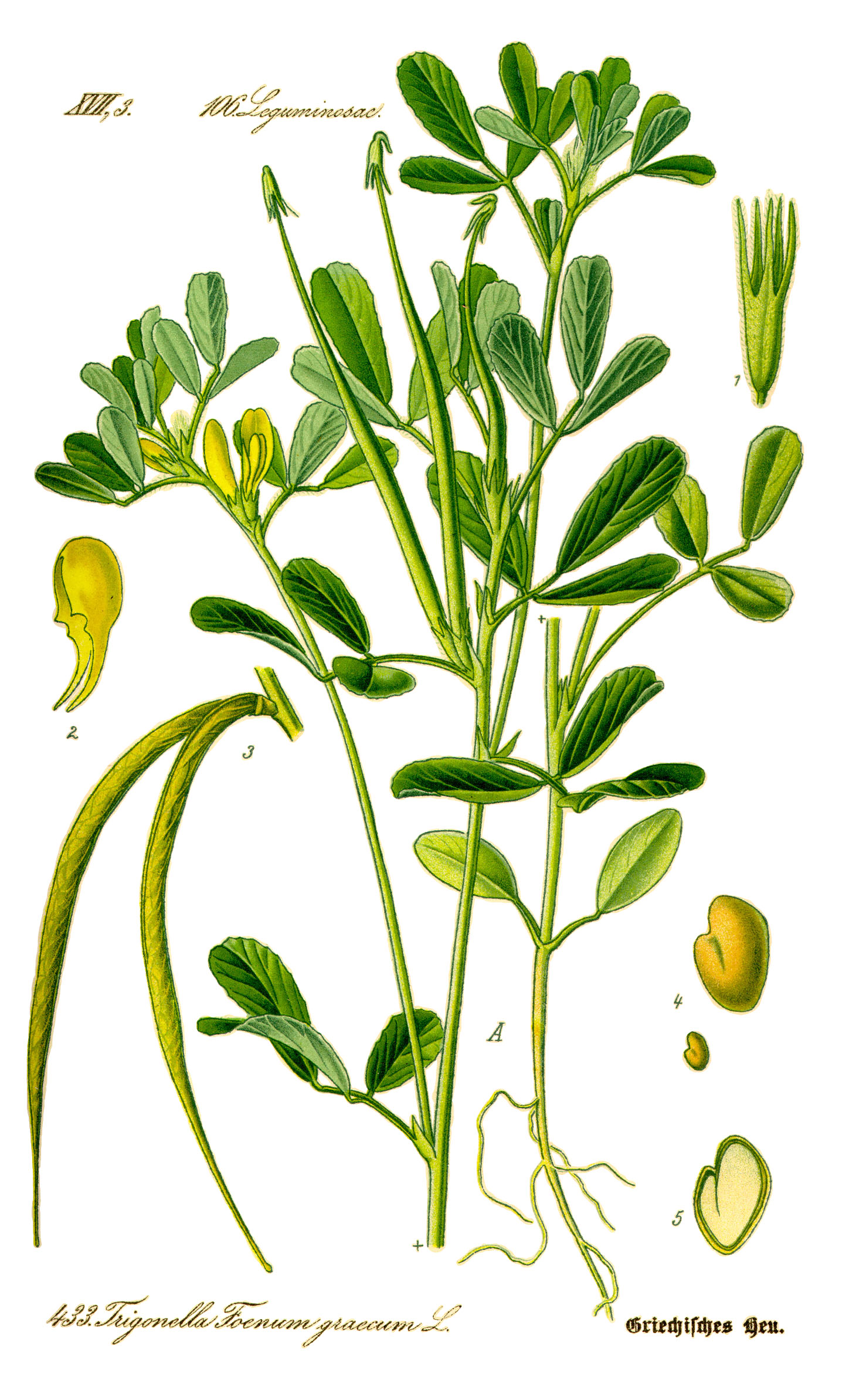
Fenugreek
Also known as: Alholva, Bird's Foot,
Bockshornklee,
Bockshornsame, Chandrika, Egypt Fenugreek, Fenogreco,
Fenugrec, Foenugraeci Semen,
Foenugreek, Greek Clover, Greek
Hay, Greek Hay
Seed, Hu Lu
Ba, Methi, Methika,
Medhika, Sénégrain, Sénégré,
Trigonella, Trigonella Foenum, Trigonelle, Woo Lu Bar. Scientific Name: Trigonella foenum-graecum; Trigonella foenugraecum. Family: Fabaceae/Leguminosae.
People Use This For: Orally, fenugreek is used for diabetes, loss of appetite, dyspepsia, gastroesophageal reflux disease (GERD), gastritis, constipation, atherosclerosis, hyperlipidemia, and for stimulating lactation. Fenugreek is used orally for kidney diseases, beriberi, hernia, and impotence and other male problems. Fenugreek is also used orally for fever, mouth ulcers, boils, bronchitis, cellulitis, tuberculosis, chronic coughs, chapped lips, baldness, and cancer. Topically, fenugreek is used as a poultice for local inflammation, myalgia, lymphadenitis, gout, wounds, leg ulcers, and eczema. In foods, fenugreek is included as an ingredient in spice blends. It is also used as a flavouring agent in imitation maple syrup, foods, beverages, and tobacco. In manufacturing, fenugreek extracts are used in soaps and cosmetics.
Safety: LIKELY SAFE ...when used orally in amounts commonly found in foods. Fenugreek has Generally Recognized as Safe (GRAS) status in the US (5). POSSIBLY SAFE ...when used orally in medicinal amounts ( 8, 13, 16, 17, 12). CHILDREN: POSSIBLY UNSAFE ...when used orally; avoid using. Fenugreek tea has caused loss of consciousness and unusual body odour in children. The body odour may be confused with maple syrup disease (12). PREGNANCY: LIKELY UNSAFE ...when used orally in amounts greater than those found in foods because of its potential oxytocic and uterine stimulant activity (18). Consumption of fenugreek just before delivery may cause the neonate to have an unusual body odour, which could be confused with maple syrup disease. It does not appear to cause long-term sequelae (11). LACTATION: Insufficient reliable information available; avoid using. Although fenugreek is used to promote lactation, there are no clinical studies testing its safety in mother or infant (22).
Effectiveness: INSUFFICIENT RELIABLE EVIDENCE to RATE Diabetes. Consuming fenugreek, mixed with food during a meal, seems to reduce postprandial blood glucose levels in patients with type 1 or type 2 diabetes. It may be given in combination with guar gum or by itself (15, 16, 21). Muffins made from a batter consisting of foxtail and barnyard millet, in combination with legumes and fenugreek, do not produce a substantial increase in postprandial blood glucose in diabetic patients (14). Gastroesophageal reflux disease (GERD). Clinical research shows that taking a specific fenugreek product (FenuLife, Frutarom Belgium), 2 grams twice daily 30 minutes before the two biggest meals of the day, significantly improves symptoms of heartburn after one week of treatment and continuing through the 2-week study period. Fenugreek was as effective as taking ranitidine 75 mg twice daily (23). Hypercholesterolemia. There is conflicting evidence about the use of fenugreek for lowering serum cholesterol (8, 13, 16). Hypertriglyceridemia. Preliminary clinical research suggests fenugreek might lower triglycerides in people with type 2 diabetes (21). More evidence is needed to rate fenugreek for these uses.
Mechanism of Action: The applicable part of fenugreek is the seed. The active constituents include trigonelline, 4-hydroxyisoleucine, and sotolon (7, 20).Fenugreek seeds have a distinctive bitter taste and odour. Sotolon is frequently used as a flavoring for artificial maple syrup (20). Soaking fenugreek seeds overnight and washing the seeds in water can decrease some of the taste and odor (13). Fenugreek seeds contain about 50% dietary fiber and pectin and may affect gastrointestinal transit, slowing glucose absorption. About 80% of the total content of free amino acids in the seeds is present as 4-hydroxyisoleucine, which appears to directly stimulate insulin (10, 19, 20, 21). This effect is glucose dependent and only occurs in the presence of moderate to high glucose concentrations. Some evidence suggests the seed consumption might decrease calcium oxalate deposition in the kidneys (4). Fenugreek contains coumarins and other constituents that might affect platelet aggregation, but this might not be significant clinically (7). Preliminary research suggests fenugreek has stimulating effects on the uterus, intestine, and heart (18).
Adverse Reactions: Orally, fenugreek can cause diarrhoea, dyspepsia, abdominal distention, and flatulence (2, 21). With large doses, hypoglycemia is possible (1). Fenugreek can cause allergic reactions including nasal congestion, hoarseness, persistent coughing, wheezing, facial angioedema, and shock (3). The paste of fenugreek applied to the scalp can cause allergic symptoms, including head numbness, facial swelling, and wheezing (3). Consumption of fenugreek by pregnant women just before delivery may cause the neonate to have an unusual body odor, which may be confused with maple syrup disease. It does not appear to cause long-term sequelae (11). This unusual body odor may occur in children drinking fenugreek tea. Loss of consciousness may also occur in children drinking tea made from fenugreek (12).
Interactions with Herbs & Supplements: ANTICOAGULANT/ANTIPLATELET HERBS AND SUPPLEMENTS: Concomitant use of herbs that have constituents that might affect platelet aggregation could theoretically increase the risk of bleeding in some people (6, 7, 8). These herbs include angelica, clove, danshen, garlic, ginger, ginkgo, red clover, turmeric, and others. HERBS AND SUPPLEMENTS WITH HYPOGLYCEMIC POTENTIAL: Theoretically, fenugreek might have additive effects with herbs that decrease blood glucose levels (15, 16). Herbs with hypoglycemic potential include devil's claw, fenugreek, guar gum, Panax ginseng, and Siberian ginseng.
Interactions with Drugs: ANTICOAGULANT/ANTIPLATELET DRUGS <<interacts with>> FENUGREEK Interaction Rating = Moderate Be cautious with this combination. Severity = High • Occurrence = Possible • Level of Evidence = B There is some concern that fenugreek might have additive effects when used with anticoagulant or antiplatelet drugs, resulting in increased risk of bruising and bleeding. Some of the constituents in fenugreek have antiplatelet effects, although these might not be present in concentrations that are clinically significant (6, 7, 8). Some drugs with anticoagulant or antiplatelet effects include aspirin, clopidogrel (Plavix), nonsteroidal anti-infl ammatory drugs (NSAIDs) such as diclofenac (Voltaren, Cataflam, others), ibuprofen (Advil, Motrin, others), naproxen (Anaprox, Naprosyn, others), dalteparin (Fragmin), enoxaparin (Lovenox), heparin, and others.
ANTIDIABETES DRUGS <<interacts with>> FENUGREEK Interaction Rating = Moderate Be cautious with this combination. Severity = Moderate • Occurrence = Probable • Level of Evidence = B Fenugreek may reduce blood glucose levels (15, 16) and might have additive effects on glucose levels when used with antidiabetes drugs. Monitor blood glucose levels closely. Medication dose adjustments may be necessary. Some antidiabetes drugs include glimepiride (Amaryl), glyburide (DiaBeta, Glynase PresTab, Micronase), insulin, pioglitazone (Actos), rosiglitazone (Avandia), and others.
WARFARIN (Coumadin) <<interacts with>> FENUGREEK Interaction Rating = Moderate Be cautious with this combination. Severity = High • Occurrence = Possible • Level of Evidence = D Fenugreek might have additive effects with warfarin and increase the international normalized ratio (INR). Some fenugreek constituents have antiplatelet effects, although these might not be present in concentrations that are clinically significant (7, 8). Fenugreek in combination with boldo has been associated with increased INR in a patient taking warfarin (6).
Interactions with Foods: ALLERGY TO FABACEAE: Chickpea, also a member of the Fabaceae family, has shown cross-reactivity in patients allergic to fenugreek. Theoretically, patients who are allergic to other Fabaceae plants including soybeans, peanuts, and green peas might also be allergic to fenugreek (3). Interactions with Lab Tests: BLOOD GLUCOSE: Fenugreek can lower blood glucose and test results (15, 16). URINE ODOUR: Fenugreek can cause a maple syrup odour in urine. Avoid confusion with "maple syrup urine" disease (9). Interactions with Diseases or Conditions: DIABETES: Fenugreek can alter blood sugar control in people with diabetes (15, 16). Blood glucose levels should be monitored closely. KIDNEY STONES (Nephrolithiasis): Theoretically, fenugreek can decrease calcium oxalate deposition and stone formation (4)
Dosage/Administration: ORAL: For diabetes, fenugreek 10 to 15 grams per day, as a single dose or in divided doses, with meals has been used (15, 16). A hydroalcoholic extract of fenugreek seeds 1 gram per day has also been used (21). For hyperlipidemia, 0.6 to 2.5 grams of fenugreek 2 times daily with meals has been used. It may be used alone or in combination with guar gum and other plant fibres (8, 16). For gastroesophageal reflux disease (GERD), a specific fenugreek product (FenuLife, Frutarom Belgium) 2 grams twice daily, 30 minutes before the two biggest meals of the day, has been used (23). Editor's Comments: The taste and odor of fenugreek resembles maple syrup, and it has been used to mask the taste of medicines (9). Fenugreek leaves are eaten in India as a vegetable (3)
Specific References: Fenugreek 1. Madar Z, Thorne R. Dietary fiber. Prog Food NutrSci 1987;11:153-74. 2. Sharma RD, Raghuram TC, Rao NS. Effect of fenugreek seeds on blood glucose and serum lipids in type I diabetes. Eur J Clin Nutr 1990;44:301-6. 3. Patil SP, Niphadkar PV, Bapat MM. Allergy to fenugreek (Trigonella foenum graecum). Ann Allergy Asthma Immunol 1997;78:297-300. 4. Ahsan SK, Tariq M, Ageel AM, et al. Effect of Trigonella foenum-graecum and Ammi majus on calcium oxalate urolithiasis in rats. J Ethnopharmacol 1989;26:249-54. 5. Electronic Code of Federal Regulations. Title 21. Part 182 -- Substances Generally Recognized As Safe. Available at: http://ecfr.gpoaccess.gov/cgi/t/text/text-idx?c=ecfr&sid= 78 6bafc6f6343634fbf79fcdca7061e1&rgn=div5&view= text&node=21:3.0.1.1.13&idno=21 6. Lambert J, Cormier J. Potential interaction between warfarin and boldo-fenugreek. Pharmacotherapy 2001;21:509-12.
| |

 Also Known As:
Also Known As: